|
5/1/2023 0 Comments T dm1 adjuvant KATHERINE will likely form the foundation of a new standard of care for this population and increase the use of neoadjuvant therapy in HER2-positive early breast cancer,” stated lead investigator Charles E. 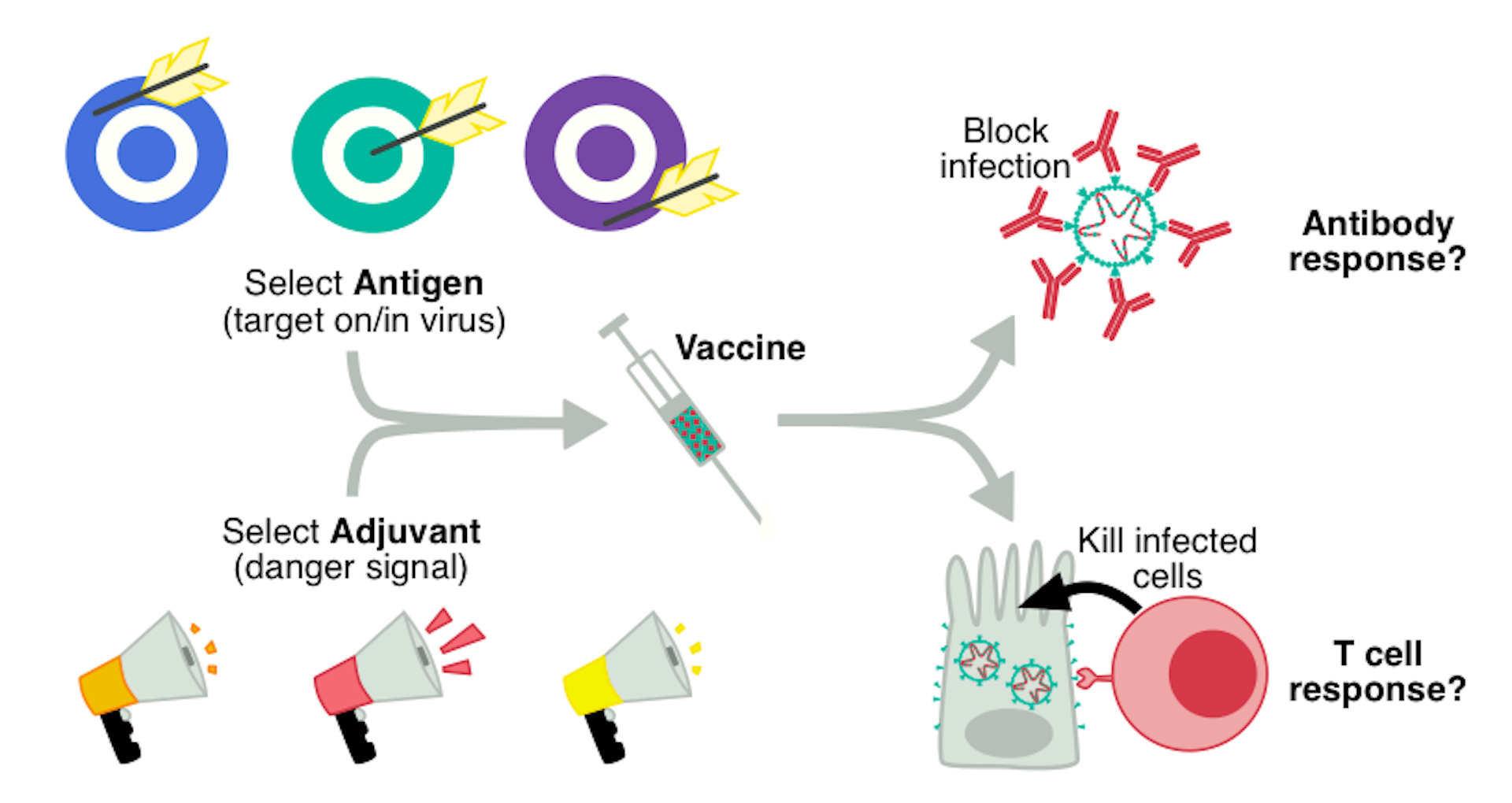
“Additional follow-up will be necessary to evaluate the effect of T-DM1 on survival. T-DM1 is approved by the FDA for the treatment of HER2-positive metastatic disease as second-line therapy after chemotherapy and HER2-targeted therapy. The benefit of T-DM1 over trastuzumab was consistent among all patient subgroups, and although there were more frequently reported adverse events with T-DM1, drug safety was manageable. The rate of 3-year survival without invasive disease events was 88% in patients who received T-DM1 versus 77% in patients who received trastuzumab. T-DM1 reduced the 3-year rate of invasive disease-free survival by 11% compared with trastuzumab.  
As a result, T-DM1 may become a new standard of care in this setting. The adjuvant use of the antibody-drug conjugate trastuzumab emtansine (T-DM1) led to a clinically meaningful and statistically significant improvement in invasive disease-free survival versus trastuzumab in patients with HER2-positive early breast cancer and residual invasive disease, despite the use of neoadjuvant chemotherapy plus HER2-targeted therapy, according to the preliminary results from the phase 3 KATHERINE trial reported at the 2018 San Antonio Breast Cancer Symposium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
